Carbon Sinks - Visualization of Quantities
-
Climate Change and Global
Warming
Carbon is in a constant state of flux being contained in any number of complex organic molecules and much simpler molecules such as carbon dioxide and methane. The atmosphere is the way by which it is transported from one place to another, here we envisage the amount of carbon in various "sinks" around the world, as well as in and above it.

There is estimated to be over 5 x 1022g of carbon on earth. One tonne is 106g, so this is 5 x 1016 tonnes, or to put it another way:
There are 50,000,000,000,000,000 tonnes of carbon on earth
About 99% of this is locked away in
sedimentary rocks such as limestone and chalk (both
of these are made of calcium carbonate CaCO
| Carbon reservoir |
Mass in tonnes of carbon x 109 (billions of tonnes of carbon) |
% of total |
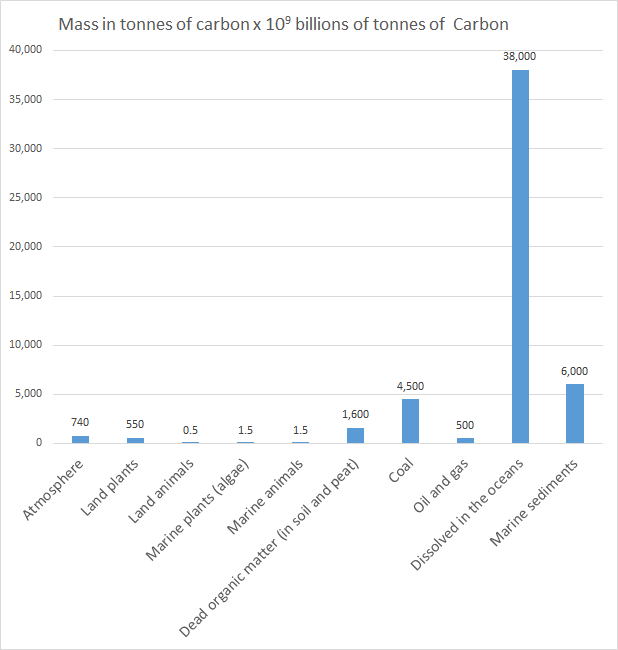
| Atmosphere | 740.0 | 1.43 |
| Land plants | 550.0 | 1.06 |
| Land animals | 0.5 | 0.001 |
| Marine plants (algae) | 1.5 | 0.003 |
| Marine animals | 1.5 | 0.003 |
|
Dead organic matter (in soil and peat) |
1600.0 | 3.08 |
| Coal | 4500.0 | 8.67 |
| Oil and gas | 500.0 | 0.96 |
| Dissolved in the oceans | 38 000.0 | 73.2 |
| Marine sediments | 6000.0 | 11.6 |
| Totals | 51 893.5 | 100 |
This distribution is rather surprising at first sight, notably because there is so much carbon dioxide contained dissolved in the oceans and relatively little is contained in the atmosphere. The atmosphere however is exceptionally important in that it is the transfer medium between the other sinks and while relatively little stays in the atmosphere a great deal passes through it.
The oceans are by far the largest sink of carbon as dissolved carbon dioxide. This is medium-term storage and is in a balance with atmospheric carbon dioxide, if atmospheric carbon dioxide is removed, it will replaced by some that was dissolved in the oceans, if large amounts of carbon dioxide are released into the atmosphere, much of it will be absorbed by the oceans. The vast majority of carbon dioxide released since the start of the industrial revolution is now held dissolved in the worlds oceans if this was not the case, the atmospheric levels would have risen far more than they even have.
It is difficult to appreciate numbers like this in a relative manner by seeing them as a list, so here is another way of showing the data, numbers in billions of tonnes:
Land animals
0.5 billion tonnes of carbon
Marine plants (algae)
1.5 billion tonnes of carbon
Marine animals
1.5 billion tonnes of carbon
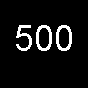
Oil and gas
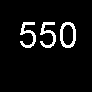
Land plants

Atmosphere

Dead organic matter
in soil and peat

Coal

Marine sediments
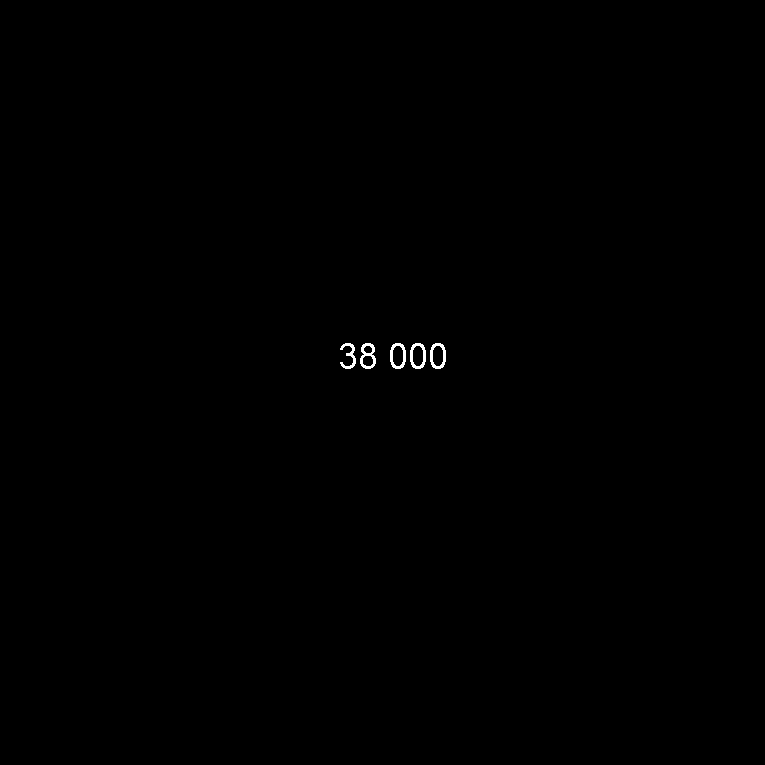 Dissolved in the oceans
Dissolved in the oceansOr as a bar chart:
Picture credits, copyright pictures used by permission: Rainforest - carbon sink - Ben Sutherland, used under Creative Commons 2.0 license
